Orion™: Whole Slide, Multiplex Immunofluorescence Imaging
Precision Spatial Biology: Orion unlocks high-plex spatial biology by providing biomarker depth and flexibility in a one-step stain and scan process
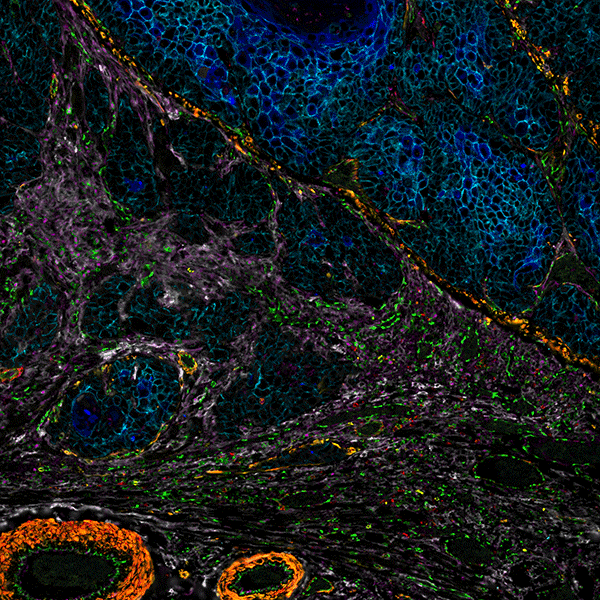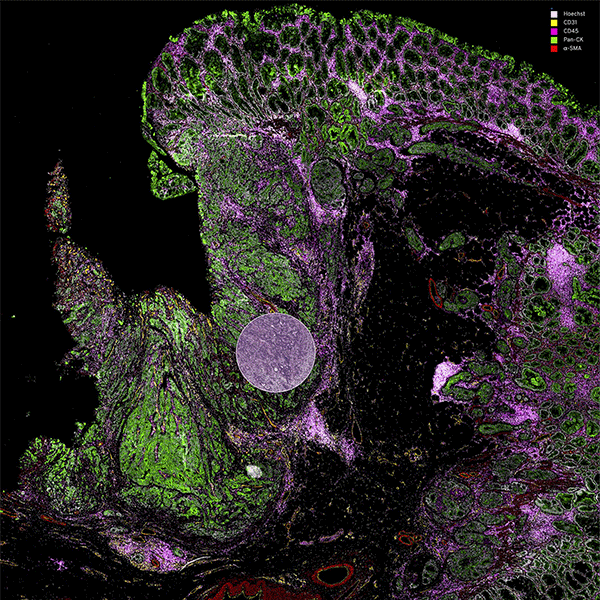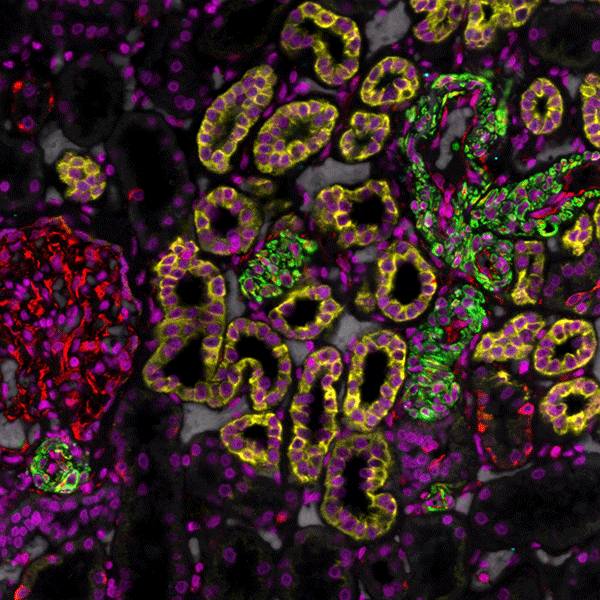
Tissue consists of heterogenous cell types, each with diverse functions and functional states, where spatial organization can impact patient health status. Resolving this complexity at the subcellular level has historically been challenged by image resolution, the number of targets that can be simultaneously assessed, and throughput.
Benefits of Orion Multiplex Immunofluorescence Imaging
Orion technology breaks these barriers, providing the fastest path to whole slide, highly multiplexed biomarker imaging data.
- High-plex, whole slide fluorescence imaging
- Stain and image in hours, not days
- Rapid, single round stain and scan
- Subcellular resolution
- Same slide H&E imaging
- Quantitative across a broad dynamic range
- OME-TIFF open file format
- Compact bench top design

 |
Nature Oncogene publication: Neoadjuvant botensilimab plus balstilimab response pattern investigated |
 |
Editorial article: Quantitative microscopy for precision spatial biology: “From samples to knowledge” |
 |
Interview: Bringing spatial biology to the clinic: “A new lens on cancer biology” |
 |
Webinar: Multimodal tissue imaging and machine learning to advance precision medicine |
 |
Webinar: Spatial analysis and high-plex immunofluorescence to study human pancreata in type 1 diabetes |
 |
Webinar: Multiplexed Tissue Imaging using the Orion Platform to Reveal the Spatial Biology of Cancer |
Orion Delivers Fast and Reliable Precision Spatial Biology
Utilizing a swift, simple workflow, all channels are acquired across the entire slide imaging in one scan while rapid data processing enables whole slide unbiased quantitation. Quick and precise sample-to-data speed provides more time for analysis and less time spent setting up your experiment.
Save even more time by using our already validated panels with the standard IHC tools you already have on your workbench.
Experience Orion:
- High fidelity data enables actionable insights
- Whole slide biomarker quantitation across a broad dynamic range
- Stain and image multiple slides per day with multiplex imaging that’s scalable and efficient
- Compatible with clinical and archived samples
- Throughput for translational and clinical studies
- Subcellular spatial context across an entire slide in just one scan

Orion utilizes flexible hardware, software, and reagents to deliver the high performance, resolution and flexibility required for research and clinical assays.
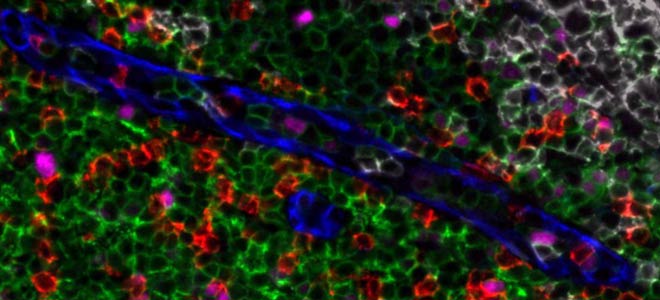
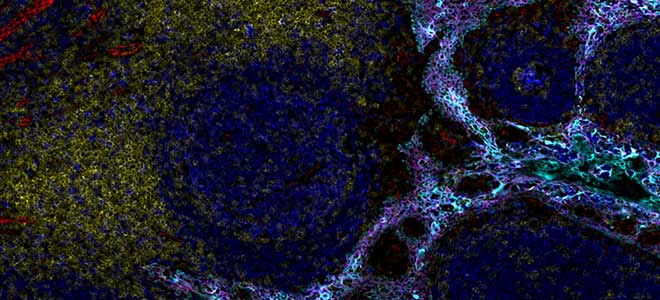
By combining speed and resolution, Orion enables comprehensive phenotypic profiling and characterization of tissue architecture, tumor heterogeneity, and the immune response for whole sections in hours, not days.
Facilitate single round immunofluorescence staining and imaging of standard FFPE or fresh frozen tissue with the additional benefits of industry standard H&E and IHC modes. Run panels from an extensive biomarker list, add in custom biomarkers, or engage our services lab for custom panels and programs.
Contact us to begin with Orion ➝
Unlock Spatial Biology with Orion for Multiplexed Immunofluorescence (MIF)
| High-plex, whole slide fluorescence imaging | Flexible panel design | ||
| Stain and image in hours, not days | FFPE and fresh frozen compatible | ||
| Rapid, single round stain and scan | Uses standard microscope slides | ||
| Subcellular resolution | Quantitative across a broad dynamic range | ||
| Same slide H&E imaging | OME-TIFF open file format |
Explore RareCyte Services and Products for Multiplex Immunofluorescence Imaging
Orion Instrument
Orion is a benchtop, high resolution, whole slide multimodal imaging instrument. A combination of quantitative immunofluorescence and bright field imaging enables robust spatial biology studies and biomarker quantitation.
Learn more about the Orion Instrument
Orion Reagents
Orion Reagents are tailored to streamline the development of custom, multiplex tissue panels within the Orion platform workflow. These reagents are compatible with formalin-fixed paraffin-embedded (FFPE) or frozen tissue sections.
Learn more about Orion Reagents

 I would like more information
I would like more information