The new standard for spatial biomarker development
Orion delivers the plex, throughput, and data quality required to translate spatial insights into clinical impact.
Advancing Precision Biology with Breakthrough Platforms
At RareCyte, we help researchers break barriers and accelerate discoveries with cutting-edge spatial biology
and liquid biopsy solutions. Our advanced platforms and expert services deliver high-resolution insights,
ultra-sensitive detection, and seamless end-to-end support—so you can get faster, more precise results.
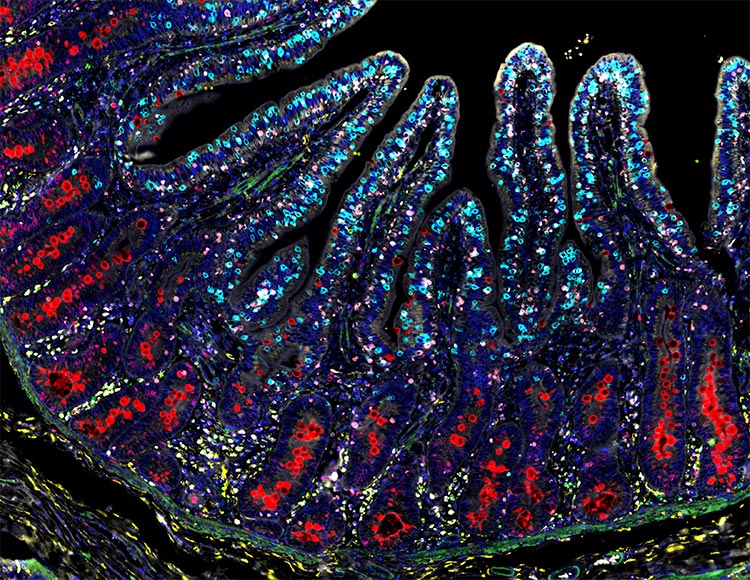
The 2026 RareCyte calendar winners are in!
RareCyte would like to thank the researchers that submitted
images for this year's calendar contest.
We are privileged to showcase these wonderful images in our annual calendar.
Spatial Biology Platforms & Precision Biology Services
Spatial Biology

High-resolution insights for tissue-based discoveries
Liquid Biopsy

Non-invasive, ultra-sensitive rare cell detection
Services

Custom assay development, clinical trial testing, and more – powered by industry-leading spatial biology and liquid biopsy platforms.

Orion™: High-Plex Immunofluorescence
Orion overcomes spatial biology challenges with 20-channel multiplex imaging, flexible panel design with validated and custom antibodies, and high-throughput whole-slide scanning to accelerate large-scale, high-quality translational research enabling actionable spatial insights.
- 20 channels per round for comprehensive spatial profiling
- Flexible panel design with validated antibodies
- High-throughput for large cohort studies or multi-user settings
Orion advances multiplex immunofluorescence into a new era.
CyteFinder®: Rare Cell Detection and Retrieval
Automate high-throughput imaging and retrieve single cells or tissue micro-regions with ease. CyteFinder delivers full automation and an end-to-end workflow—from blood collection to single-cell isolation—ensuring sensitive, accurate, and reproducible results.
- Easy-to-use staining panels for identification and classification of rare cells
- Accelerated analysis and improved review concordance with advanced CTC machine learning algorithm
- Blood collection to CTC characterization tailored for high-throughput laboratories and access to a global CRO network

Dedicated Lab Services
RareCyte provides high-quality data for CTC applications and spatial biology, supporting therapy selection, disease monitoring, global research and clinical trials. Our expert team offers flexible, milestone-based support to align with your timelines and budget – so you can advance your work efficiently.
- Custom assay development
- Clinical trial testing, analysis, and reporting
- Best-in-class platforms, QMS, and CLIA laboratory
- Dedicated project team focused on Quality, Fast Turnaround Times, and Responsiveness
Conferences & Webinars
Conference:
NextGen Omics, Spatial & Data US
Mar 31 - Apr 1, 2026
RareCyte team:
Jim Gallagher, Tad George
Conference:
American Association for Cancer Research
Apr 19-22, 2026
RareCyte team:
Xiao Chen, Melinda Duplessis, Jeff Chamberlain, Tad George
On Demand:
Bridging the Discovery-to-Translational Gap in Spatial Biology
Available Now
Speaker:
Zhihong Chen, PhD
CTO, OCCAM Immune, at Icahn School of Medicine, Mount Sinai Hospital, New York
On Demand:
Mastering Multiplex IF: Easy Strategies for Reliable Spatial Biology Results
Available Now
Speaker:
Simon Goldstein
Microscopy Specialist II
La Jolla Institute for Immunology
RareCyte in the News
Article:
Optimizing spatial proteomics for discovery and translational research

Tad George
Senior VP of Biology
RareCyte
Article:
Advanced spatial biology sheds new light on therapeutic research

Dr. Zhihong Chen
Chief Technology Officer
OCCAM Immune
Nature Oncogene publication:
Neoadjuvant botensilimab plus balstilimab response pattern investigated

Dr. Pashtoon Kasi
Weill Cornell Medicine
