Videos
| Precision Medicine vs Persuasion Medicine: Measuring vs Reading HER2 |
| Yale Cancer Center Grand Rounds with Dr David Rimm |
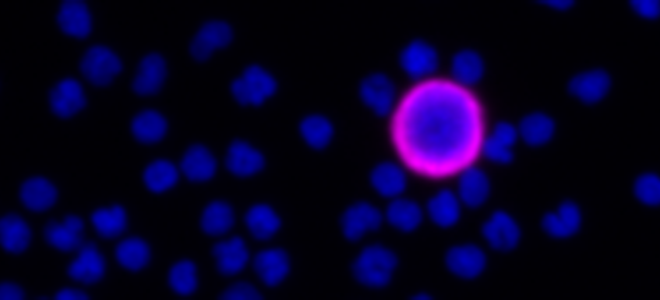
| RarePlex® Panel and Developer Assays for CTC enumeration and characterization |
| Presented by Arturo Ramirez, Ph.D., Director of Oncology R&D This 7 minute overview of RarePlex Panel Kits and Developer technology highlights available circulating tumor cell assays from RareCyte and presents an introduction to our assay design and validation. |
| Working with the RarePlex® Developer Kit |
| Presented by Edward Lo, Ph.D., Scientist, Assay Development Lead Developer technology from RareCyte enables you to add up to two custom biomarkers to your CTC assay. This 5 minute video will walk you through each step of the Developer process and provide examples of completed assays. |
| RarePlex® Assays: Design and Analytical Validation |
| Presented by Dr. Tad George, Sr. VP of Biology R&D RarePlex Assays are sensitive, specific, and reproducible assays for CTC detection and biomarker expression. Gain insight into the principles RareCyte applies to its assay design and validation process in this 11 minute video. |
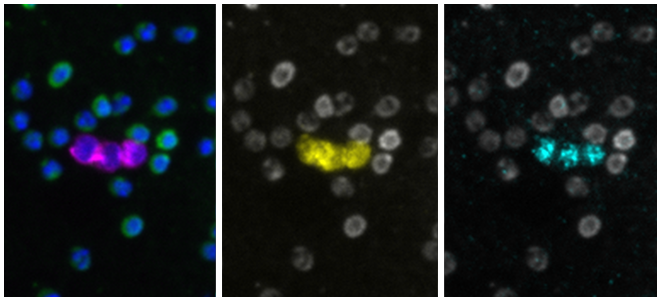
| Tissue transcriptomics with CyteFinder® II Instruments |
| Presented by Jennifer Chow, PhD, Principal Scientist This 5 minute overview of RareCyte's spatial transcriptomics technology highlights how multiplex imaging and needle-based retrieval with the CyteFinder II platform enables discovery applications with FFPE and fresh frozen tissue samples. |
| RareCyte technology presentation at Harvard Medical School |
| RareCyte technology presentation at Harvard Medical School titled "High Performance Multiplexed Imaging and Microregion Retrieval for Sequencing" presented by Leighton Howells, Vice President of Business Development, and Kyla Teplitz, Sr. Product Manager. |