Unlock Spatial Biology with Orion Technology: Explore the possibilities for research and clinical applications
Rarecyte: Precision Biology for Life Sciences
Welcome everybody. So very briefly a few words about RareCyte. RareCyte is an American company founded in 2009 and we are based in Seattle, and we are a precision biology company developing high performance microscopes dedicated to pathology type samples, all types of biopsies including liquid and tissue biopsies.
At RareCyte, we focus on two main areas: first the rare cell analysis and retrieval of the cells it’s mainly used for the CTC the circulating tumor cells and second the fast growing field of spatial biology and its highly multiplex tissue analysis.
So today I will talk about the Orion which is a really high speed highly multiplex spatial analysis imaging platform, and if some of you are interested, contact us after the webinar and I will send you a few words about the CyteFinder II instrument here on the left which includes beside the imaging part, an onboard device for the microregion retrieval to collect the cells of interest for downstream genomic analysis.
Unlocking Spatial Biology with Orion Technology
So first of all why did we develop the Orion system? So as you know the tissue consists of heterogeneous cell types, where cells have diverse functions and functional states. The spatial biology goal is to understand which cells are present, where they are localized in the tissue but also the pattern of biomarkers expression ideally at the cellular level. The goal is also to understand how the cell is organized and how they interact to influence the tissue microenvironment. So resolving this complexity is challenged by several things. Challenged by the autofluorescence of the cell and may be challenged by the overlaps of the different fluorescent signals which limit the number of targets that can be simultaneously evaluated. It’s also challenged by the very low throughput, usually due to a very long multi-cycle staining procedure and also by a very slow acquisition time.
Desirable Design Features of Tissue Imaging Platform Compatible with Pathology Workflows
When we have asked pathologists and researchers to list the characteristics of the optimal tissue imaging platform compatible with pathology workflows, this is the feedback we have received. I’m not going to read all of this in detail now, I just want to highlight here some keywords. We can see 10 or plus simultaneous markers, whole slide, rapidity, high resolution, to have panels and flexible choice etc and it’s within this frame that we have developed the Orion platform. It includes hardware and software combined with a strategic fluorophores palette to enable rapid, high resolution, whole slide spatial biology results via a practical single cycle process.
Orion: Spatial Biology Realized with Single Step Staining
So from a high level, Orion has a very straightforward workflow. Essentially you prepare your slide on traditional microslides, with normal tissue sections. We are working mostly with FFPE preparation. I think what’s important here is that first Orion analysis is done on the whole slide and second the staining is actually a single step staining process which is done with really standard immunofluorescence protocol with all the different biomarkers addressed at the same time. It takes only one scan for all the fluorescent channels and then the data can be analyzed downstream either on the onboard qualitative software named Artemis or on the third-party software.
High Plex Imaging in Hours, Not Days
The question is how do we do this? So well we are still using fluorescence because fluorescence is really needed for the high speed we want to achieve. In short, we image roughly one square centimeter and we can do that in a little bit less than an hour and a half so it’s very fast. Traditional fluorescence imaging as you know use a single dye in each spectral region of the entire spectrum and that generally speaking allows for a maximum of four or five markers to be viewed at the same time. Some people use an alternative approach here represented at the top of the screen. So they use an alternative approach to increase the number of markers they perform sequential or iterative immunostaining by repeating many cycles of staining, imaging, stripping, washing, and then again the staining, imaging etc. With the high risk to damage the tissue, with the difficulties to stack the different images acquired at each cycle and again with the very low throughput associated to this method. But with the Orion technology here at the bottom of the screen, we extract specific signals from dyes that are closely adjacent in the spectrum which allows to access multiple markers for spectral region.
So with Orion we leverage the fluorescence in a smart way. We are doing all the colors at once and then we resolve the spectral overlaps by using the powerful algorithm to perform a deconvolution.
High Resolution, Multiplexed Imaging of Key Cellular Markers
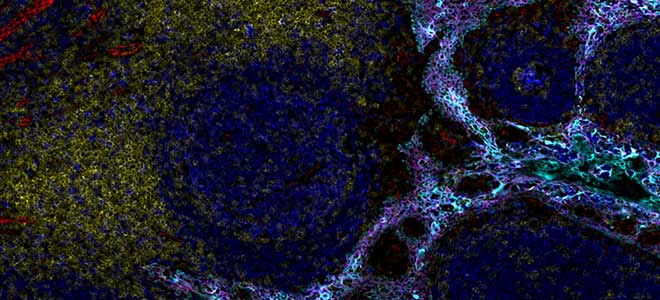
To illustrate this we can take this example, this section of lung is showing five key biomarkers: cytokeratin, CD68, and three T-cell markers. The five antibodies used here are conjugated to five dyes which are all from the same region of the spectrum. It’s the orange region so around 600 nanometers. Orion is able to resolve all this signal, so each individual biomarker can be visualized independently. That’s what you can see along the bottom of the slide. With no intervention from the user the cellular complexity becomes resolved. Keep in mind that here for this example we have done this for a single spectral region, but now with Orion you multiply this across the entire spectrum and so you increase a lot of possibilities for multiplexing.
Customizable Orion Panels
We have also optimized some panels, for example our immuno-oncology 12-plex panel contains the typical immune response markers and also some specific checkpoint markers. And here on the left side you can see our immuno-response 9-plex panel which is giving a more general immune response. Those two core panels, they offer some flexibility in order to include custom biomarkers of your choice such as architectural markers, cancer specific biomarkers but it could also be a marker of the cellular cycle or specific of a neuronal disease for example.
Whole Slide High-plex Imaging
As I mentioned earlier Orion is able to scan the whole slide so here is an example. This is a 17-plex panel applied on a tonsil section. We have scanned here a little bit more than one square centimeter of tissue. It took less than two hours to image everything in the 17 channels. The image here is showing the architecture from a high level. But of course Orion is offering very high resolution as well. Most people who use the system they use our 20X objective which gives about 0.5 micron resolution. it is possible to get 4X, 10X, 20X, and 40X magnification. Here it’s 20X, you can see down into the different subsets and the high level resolution over the whole slide and here we have zoomed in on five different areas of this tonsil section.
Tonsil | 17-plex
To help with the visualization and the understanding of these 17 biomarkers staining it’s easier to look at combinations of given biomarkers. Here we have four times the same region which is displayed with four different combinations of markers which we are going into detail in the next slide as you can already notice, the very good resolution here allows us to really have a look at the phenotype at the single cell level.
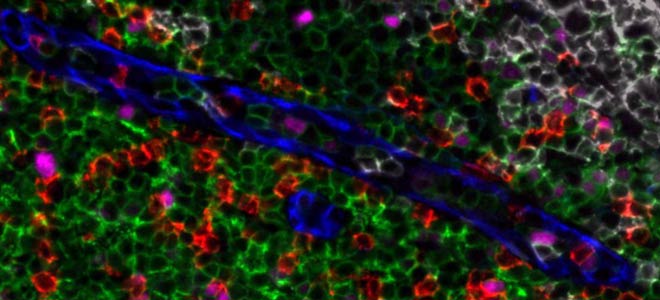
So here we see a B-cell follicle with most of the proliferating B cells located in the germinal center. So they are CD20+ in blue and PCNA+ in red. The CD4 lymphocytes in yellow are mainly confined to the inter-follicular T-cell zone. As for the macrophages which are in magenta, they are mainly concentrated within the follicle and are relatively few in the inter-follicular T cells zone.
In this second image we confirm with Ki67 in yellow and PCNA in red that the proliferation of the B cells is intense within the germinal center. The PD1+ cells in light blue, they do not proliferate which is consistent with the presence of this immune checkpoint inhibitor, and around the CD3+ lymphocytes are found in the interfollicular zone.
Here on this third image, we have zoomed in on the inter-follicular T-cell zone and we can see there the expression of FOXP3 in red on one subtype of T lymphocytes, so on the CD4 which are in light blue, but not on the CD8 which are in pink here. I would like to take just a few seconds to remind you that with Orion we work with antibodies that are directly conjugated. There is no need of whatever strategy of signal amplification, no secondary or even tertiary leveling, no tyramide signal amplification. So using directly conjugated antibodies Orion enables here to collect a very clear image of an intracellular marker such as FOXP3 for example, which is expressed quite weakly and this is possible thanks to the use of seven high powered lasers installed inside the Orion instrument, and thanks to a specific algorithm to treat the signal.
Finally, on this last image we are focusing on the myeloid cells we see a co-expression of PD-L1 and CD68 in red and in yellow on the macrophages of the germinal center. And the M2 macrophages which are CD163+ in light blue are excluded from the germinal center. We can see them actually right here and the last image on the right at the bottom.
Access Any Sample
So with Orion we can assess any kind of sample; lung, melanoma, tonsil, thymus, pretty much any kind of tissue that you can put on a slide will work well with the Orion system, including tumor microarrays. There is no problem at all to scan this type of sample.
Track Disease State
Back to another example using the same panel, the 17-plex. Here applied on normal lymph node on the left, on tonsil during inflammation, it’s in the middle, and on the chronic lymphocytic leukemia cells in a lymph node on the right. And without going into a detailed analysis just based on the colors of the different areas, we can already see clear structural differences on the normal lymph node. So on the left the B cell follicles which are in blue contain few proliferating cells and no germinal center. The follicles are surrounded by T cells areas in white. In the tonsil inflammation so the image in the middle we have many of the B cell follicles that contain germinal centers, those red areas that you see, which is consistent with the inflammatory response. And then for the lymphoma completely on the right, clearly the proliferating B cells are dominating there are very few germinal centers and very few T cells.
It’s actually quite common for people to perform serial sections for H&E staining, the hematoxylin and eosin staining, to look first for specific areas of concern and then look deeper with the immunofluorescent staining. With the Orion workflow it’s actually feasible to get the H&E imaging of the same section; so the same exact slide can be stained. What we do is we first stain the sample for the multiplex fluorescence, do that imaging using the Orion system, remove the coverslip, and then we stain the slide again with H&E and image it again using the bright field mode of the Orion system. So we can see for the same section, the H&E staining as well as the high plex fluorescent imaging.
At this stage I would like to invite you to visit later our web page and to play with two demo data sets that we are providing. To explore the data, it’s easy just go to rarecyte.com/Orion and click on “view data se”t. The first data set is a 16-plex image of oral squamous cell carcinoma and the second is a 17-plex panel on a lung sample.
Once you have selected one of the data set, you will then be able to look at the whole slide scan and to zoom into it as you wish, and display the different color corresponding to the 16 or the 17 biomarkers.
Orion Data Analysis
Regarding the analysis with Orion, we are providing the Artemis software to visualize the image acquired. From Artemis we can export pyramidal open source format files so the analysis can be done on many software solutions already existing such as Qpath, Halo, Visiopharm but also some open source and free analysis solutions that have been developed by different research groups. Orion data can be integrated into an existing quantitative analysis pipeline without the need to modify the exported file.
Qualitative and Quantitative Evaluation of the Tissue Microenvironment by High Resolution 17-plex Immunofluorescence Reveals Distinct Cell Populations
In this example, HALO AI by Indica Labs is used for the quantitative analysis of images obtained with Orion. Three kind of samples were analyzed, normal lymph node, tonsil during inflammation and the lymphoma. Two algorithms were used here, the first one for the segmentation of the cells to separate and measure each individual cells, and the second algorithm to run a tissue classification and recognize automatically some types of tissue like the B-cell zone, the T-cell zone, the epithelium, and the stroma. Then from that, it’s possible to quantify the various complex phenotypes in each of the different areas of the tissue. I can share this poster with you, just ask me.
MCMicro Pipeline
And to give you more information on quantitative data I would like to highlight a recent publication in Nature Methods which presents MCMicro, an end-to-end processing pipeline to transform large, multi-channel, whole slide images into single cell data. Also you will see on the right-hand side of the screen a link to a webinar given by Sandro Santagata of Harvard who highlights the use of this tool, MCMicro, that they have developed, to analyze the Orion data.
Uniquely Orion: Spatial Imaging
To summarize and close my presentation of the Orion technology here are five points which I believe make Orion really a unique tool. The possibility of doing a whole slide imaging will prevent sampling errors allowing to see actual patient-to-patient variability. The straightforward single tissue staining procedure which removes experimenter bias. The speed of acquisition and the contained-cost-per-slide that enable translational and clinical studies. The possibility to image with true subcellular resolution enabling real tissue mapping. And then the possibility to image the same section in brightfield after standard tissue stains which is very important for pathology labs.
Well this is the end of the presentation. I hope you are now enthusiastic to join the world of special biology using our Orion technology and we can move now to the Q&A session. Here are our contacts.
Q&A on Spatial Analysis and Spatial Biology Technology
Thank you, Mickael, so we have had a few questions coming through the Q&A box. I’m gonna be asking you these questions and hoping that you have the right answers for the audience. So the first question that we have here is about the antibody conjugation process and especially the question is about whether it’s possible for a user to conjugate primary antibodies themselves and combine them with existing panels.
Q: Is it possible for a user to conjugate primary antibodies themselves and combine with existing panels?
Yes actually the way we are developing our strataplex reagent is, with Orion, we have a palette of fluorescent molecules that can be conjugated to your own antibodies to the antibodies of your choice to include them into the panel. So that doesn’t mean that it’s not possible to use another molecule, but as a good start I will highly recommend to use those conjugation kits. It’s very straightforward procedure to label the antibodies and then they will fit perfectly with the rest of the panel.
Thank you, Mickael. Another question is about tissue autofluorescence.
So is the Orion platform able to handle tissue autofluorescence? And a question related to that one, do we have any pre-treatment recommendation before we do the analysis or the acquisition?
So in our procedure when we prepare the slide during the staining part, just before doing the staining, we actually have a process to try to decrease a lot of the background by doing some illumination of the tissue, so and for the rest of the autofluorescence, it’s actually quite useful to have a little bit of autofluorescence of the tissue, because we can visualize that also in the final image that we have acquired. So the autofluorescence it’s actually I would say useful to see the general morphology of the tissue and so it’s always good to have a little bit of it, but yeah the purpose of the algorithm that we use and the deconvolution that we perform, it’s actually to make sure to remove all the overlaps between the different fluorescences that we are using but also we are taking care in the algorithm of the autofluorescence. So there are several steps of making the final image very clear and beautiful.
Thank you, Mickael. Another question that we have is about automated tissue staining and whether we do recommend using an autostainer for sample preparation.
With automated tissue staining, do we recommend using an autostainer for sample preparation?
Well you certainly can use autostainer for Orion samples, but it’s not essential. One of the core design features of the Orion is to remove the burden of long, multi-step tissue handling procedures by enabling a tissue staining in a single step. You can either use automated or non-automated sample preparation, whichever suits your existing workflow. But, I would say it’s not mandatory. It’s a very simple procedure, classic procedure for staining, and I would say I do not expect people to have to actually stain 40 samples at the same time, with a huge panel. So yeah it’s compatible but not mandatory.
Okay thank you very much for that.
What Specimens can be used with Orion Technology?
We have another question but I think you answered it already about the nature of the specimens that can be used with the Orion technology, mainly FFPE samples are amenable to the technology.
FFPE, frozen samples, whatever you can put on the slide I would say, we will be able to scan it with the Orion. It has to be a microscope slide.
Okay thank you another one is about the objectives.
Do we have the four mentioned objectives already pre-installed in the system or is there a selection that needs to be made before the system ships?
The question is whether we do have the four mentioned objectives already pre-installed in the system or whether there is a selection that needs to be made before the system ships?
So, I would say, the instrument comes with all the objectives, the 4x, 10x, 20x, and 40x. Now what is the recommended magnification, for instance for subcellular localization; You can use the 40x objective so it has a resolution of 0.2 microns. And then the system also has the 20x objective on board and this one gives a resolution of 0.5 microns per pixel. But yes it’s going to go to scan four times faster than the 40x. So you don’t have really to give up throughput and resolution, the throughput for the resolution. Short answer I would say the majority of the users are using the 20x because it gives very good resolution and it goes very fast. So, if we look at the process, the way the machine is dealing with the scan, you will first run a scan at 4x. Then we are using that very quick scan we are going to let know the software where we want to make the image. So using a very basic tool you just tell the system okay I want this area to be scanned and then it will start the scan using the 20x or 40x that you have selected – that’s the process.
Okay thank you Mickael.
Do we have anything right now which is ready for model specimens?
Another question is about the menu of panels that we do currently have, especially regarding the mouse model. Do we have anything right now which is ready for model specimens?
Short answer, we do not have yet panels for mouse. We have developed two panels for humans. I expect that the panel, the 9 plex panel which is the basic immuno response. It will be the one that people are going to use, and based on that very small core panel, it’s already a 9 plex, you can add your antibodies of choice or you can start from scratch. But I would expect that people always want to, at least in the immuno-oncology field, they will want to measure those markers, so why not start with a core panel and just build your personal panel on the top. But for the mouse we don’t have anything available. We don’t have a commercial product yet for that.
Okay thank you, Mickael.
Is there a loader within the system for slides?
I think we can take two more questions so another question is about the loading of the slides in the system. People are trying to understand if there is a loader within the system or if you need to load the slides one by one?
So the procedure to load is very straightforward: you click on open then you have the two holders that go out of the system and you just manually load your two slides click close and press scan. So in term of interaction with the machine it is very, very simple, there is no other action to do. To answer the question, we do not provide an autoloader or something like this so for now we didn’t work on making a system to automate this. You can load two slides into the system, one on the left and one on the right and start the scan of it. Okay thank you Mickael.
Do you offer a demo of Orion?
And the final question is about the possibility of demoing the Orion instrument in my lab. Essentially, people are trying to understand if we do offer a demo of the system?
So, the question is about a demo instrument in the lab. So right now, we do not engage on, in on-site Orion demonstration. However, we do offer proof of concept studies to support our collaborators so if it helps for the decision-making process, so if people want to discuss that type of demonstration, don’t hesitate to contact us, so we can have a talk about it. But yeah we do proof of concept studies.
Thank you Mickael.
You’re welcome.Well that concludes our webinar for today. I hope you enjoyed it if we did not get your question. We will be following up via email in the next couple of days, so do expect that and have a great rest of your day. Great afternoon and great evening. Thank you.