Developer Kits – 405/488
Download Specification Sheets
Download Test Menu Catalog
RarePlex® AR/ARv7 CTC Panel Kit
Prostate cancer is the second most common type of malignancy and one of the leading causes of cancer deaths in men worldwide.1,2 Since androgen-mediated signaling is the predominant driver of prostate cancer proliferation3 the androgen receptor (AR) is an important therapeutic target.
Therapeutics have been developed that target the ligand-binding domain of the AR protein.4 It has been determined that some patients may not benefit from these treatments or may develop secondary resistance since they do not express this ligand-binding domain.
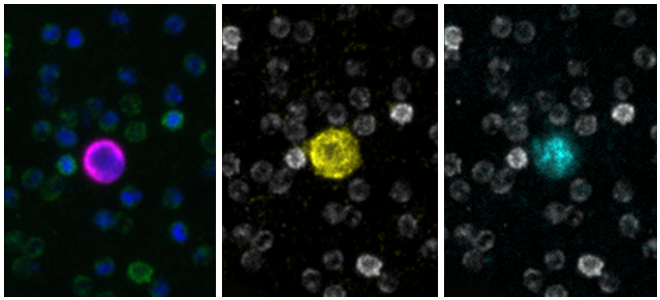
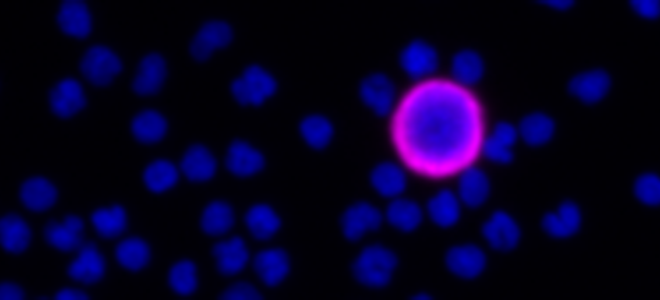
Detection in patients’ circulating tumor cells (CTCs) of androgen receptor splice variant 7 (ARv7), lacking the ligand-binding domain, shows a link with treatment failure. Thus, identification of ARv7 in CTCs could help determine if a patient has become resistant to AR-targeted therapies. As prostate cancer is a rapidly evolving field where studies identifying and characterizing new biomarkers aim to identify a cancer-halting treatment, assay flexibility is essential to keep pace with novel discoveries.
RareCyte’s RarePlex AR/ARv7 CTC panel kit (assay code 0904-LB) provides highly accurate, repeatable, and precise results for CTC count and AR and ARv7 biomarker expression; and is suitable for use in large, multi-center clinical trials of prostate cancer.
Benefits of the AR/ARv7 CTC assay:
- Simple processing of blood to slides for sample banking
- Convenient pause points in the workflow enables sample control and transport from remote collection sites
- Flexible sample handling – processing and analysis may be performed at the same site or at a separate clinical research laboratories
- Extensive validation and a highly automated workflow yield accurate and highly reproducible results
- Worldwide network of contract research organizations (CROs) equipped to run the workflow in support of global clinical trials
Additional resources for the RarePlex AR/ARv7 Panel Kit
Download the specification sheet →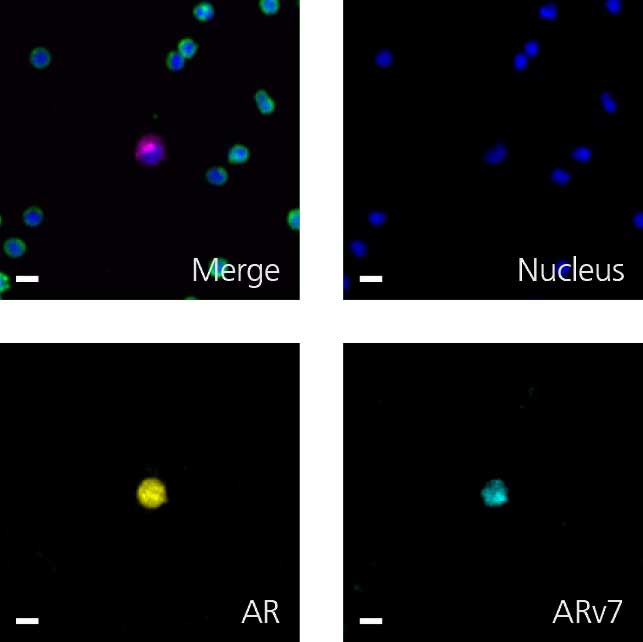
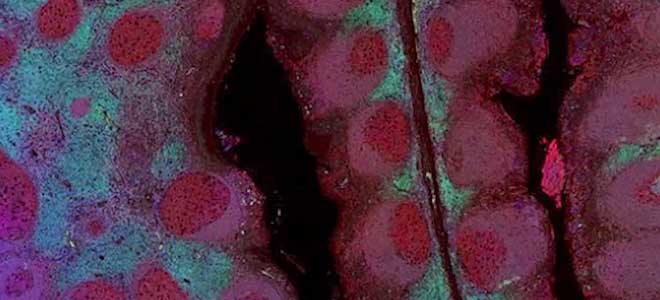
Scale bar represents 10μm.
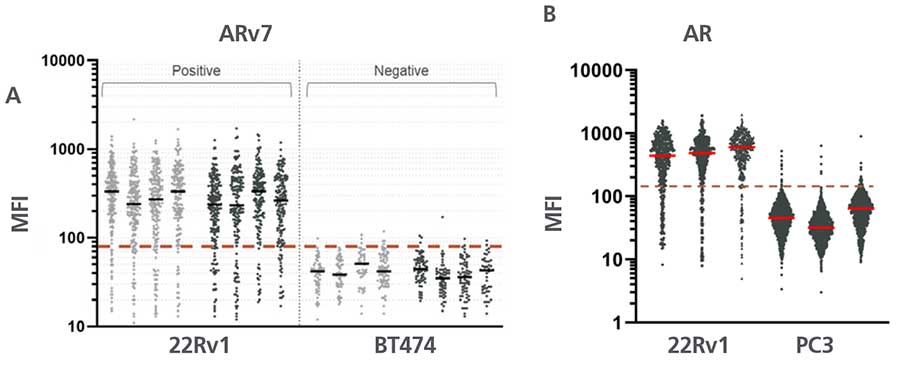
B: Per-cell AR MFI obtained for 22Rv1 and PC3 spike in slides. Each group represents data from 4 slides stained on one stainer run with the 0904 AR/ARv7 (dark) assay. Median values represented red lines. Optimum threshold for positivity is 120 for AR, represented with dashed line.
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [published correction appears in CA Cancer J Clin. 2020 Jul;70(4):313]. CA Cancer J Clin. 2018;68(6):394-424. doi:10.3322/caac.21492)
- Rawla P. Epidemiology of Prostate Cancer. World J Oncol. 2019;10(2):63-89. doi:10.14740/wjon1191
- Cioni B, Nevedomskaya E, Melis MHM, et al. Loss of androgen receptor signaling in prostate cancer-associated fibroblasts (CAFs) promotes CCL2- and CXCL8-mediated cancer cell migration. Mol Oncol. 2018;12(8):1308-1323. doi:10.1002/1878-0261.12327
- Helsen C, Van den Broeck T, Voet A, Prekovic S, Van Poppel H, Joniau S, Claessens F (August 2014). “Androgen receptor antagonists for prostate cancer therapy”. Endocrine-Related Cancer. 21 (4): T105–18. doi:10.1530/ERC-13-0545. PMID 24639562